Partnership Opportunities
Elevate Your Services to Our Audience of Pulmonary Fibrosis Experts
The 8th IPF Summit provides a unique and unmissable 3-day opportunity to connect with worldwide innovative leaders and offer invaluable assistance to aid in advancing their critical candidates through the clinic.
Capitalize on this rare opportunity to showcase your solutions to our audience of highly engaged decision makers prioritizing the progression of disease treatment in pulmonary fibrosis.
We will work with you to build a bespoke agreement tailored to your business development goals, ensuring you meet who you want to meet and how you want to meet them, from whole audience presentations to 1-2-1 meetings, hosting private dinners and beyond.
What Do Pulmonary Fibrosis Drug Developers Need Your Help With?
Preclinical models which better recapitulate the lung microenvironment and disease pathophysiology and the dynamic nature of pulmonary fibrosis progression.
Advanced screening and imaging technologies enabling early detection and diagnosis, real-time monitoring of progression and efficacy of therapeutics, and more.
Comprehensive biomarker services offering a blend of established and trusted biomarkers alongside cutting-edge, minimally invasive biomarkers.
Preclinical CRO’s with specific biological understanding for target identification and validation to assist with drug discovery and design.
Clinical CRO’s with respiratory expertise and experience in the sensitive nature of clinical trial design in Idiopathic Pulmonary Fibrosis and Interstitial Lung Disease.
And Much More....
What’s In It for You?
Expand Your Network: Connect with a diverse array of industry professionals and decision makers as well as researchers and clinicians to broaden your network and explore potential collaborations beyond the conference.
Gain Industry Insights: Engage in discussions led by industry leaders to gain valuable insights into emerging trends, challenges, and opportunities in the pulmonary fibrosis landscape, to help shape your future decisions and ensure you stay ahead of the curve.
Demonstrate Thought Leadership: Position yourself as a thought leader by sharing your expertise, insights, and innovative solutions with a captive audience of industry stakeholders, reinforcing your company's reputation and credibility.
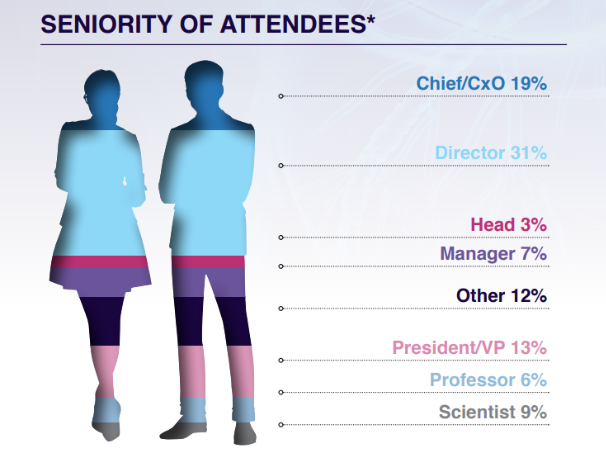
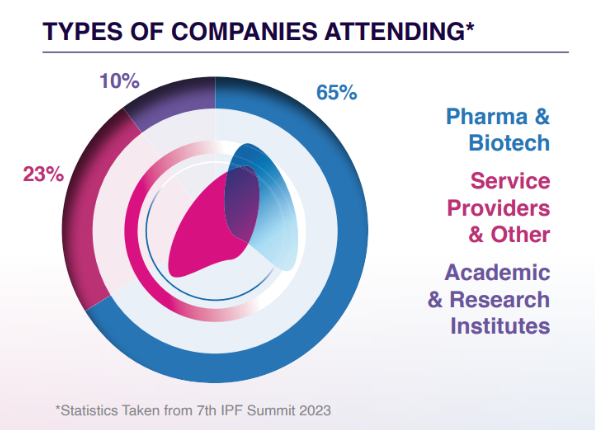
Who Will You Meet?
2024 Partners
Expertise Partner

Program Partner

Innovation Partner

Industry Partner

Exhibition Partner

Industry Partner

Exhibition Partner


